Ir/PTC cooperatively catalyzed asymmetric umpolung allylation of α-imino ester enabled synthesis of α-quaternary amino acid derivatives bearing two vicinal stereocenters†
Abstract
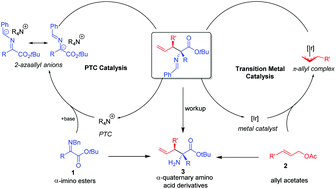
A novel Ir/PTC (phase-transfer catalyst) cooperatively catalyzed asymmetric umpolung allylation of simple α-imino esters is developed and it provides facile access to α-quaternary amino acid derivatives bearing two vicinal stereocenters. Both the metal catalyst and the phase-transfer catalyst are crucial for this methodology. The reaction features good yields, high diastereo- and enantio-selectivities, ease of scale-up to gram scale and further transformation of the products (e.g. to quaternary proline analogues with multi stereocenters).


 Please wait while we load your content...
Please wait while we load your content...