A 3-step chemiluminescence method for chemical oxygen demand measurement with dichromate oxidizing reagent
Abstract
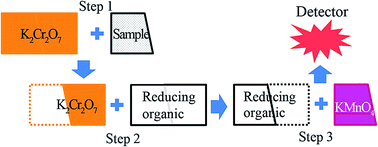
In this study, the use of potassium dichromate as an oxidizing reagent in the 3-step chemiluminescence (CL) chemical oxygen demand (COD) method, based on the principle of the permanganate COD (CODMn) method (JIS K0102), was developed (3-step CL CODCr method). The sample reacted with an excess of dichromate under heating, and the remaining dichromate was neutralized by a reducing organic compound. Then, the remaining amount of the reducing organic compound was detected in the CL detector using permanganate CL reagent. Six organic compounds – gallic acid, phenol, pyrocatechol, ascorbic acid, pyrogallol, and potassium hydrogen phthalate – were investigated as the reducing organic compounds. The CL kinetics of these compounds with acidic permanganate reagent were studied, and pyrogallol showed the highest signal with the fastest relevant response. Hence, pyrogallol was chosen as the reducing organic compound. The enhancement effects of seven metal ions – Cr(III), Mn(II), Cu(II), Ag(I), Fe(II), Fe(III), and Mg(II) – were investigated. The results showed that Cr(III), Mn(II), Fe(II), and Fe(III) enhanced the signal. Fe(II) and Fe(III) showed the highest enhancement effects, in which the signals increased to 188% and 205%, respectively. However, Fe(II) made the permanganate reagent become less stable due to the reaction between Fe(II) and permanganate and the enhancement decreased significantly with time. Hence, Fe(III) was added to the permanganate reagent at the optimum concentration of 0.45 mmol dm−3. The correlation between the 3-step CL CODCr method and the conventional CODMn for several organic compounds showed a good determination coefficient R2 = 0.9594 (n = 16). The limit of detection was 0.083 mg L−1, using gallic acid as a standard. The method was applied to measurement of river water samples, obtaining results in good correlation with the CODMn method.


 Please wait while we load your content...
Please wait while we load your content...