Detection of total bile acids in biological samples using an indirect competitive ELISA based on four monoclonal antibodies
Abstract
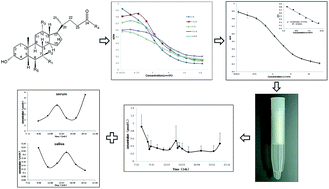
Total bile acids (TBAs) are sensitive indicators of hepatic injury, neonatal jaundice, and other metabolic diseases, and an enzyme cycling method has been used in clinical practice for the determination of TBA concentration for more than 20 years. However, this method has several limitations, particularly in terms of sensitivity and practicality. Here, we investigated a newly developed indirect competitive enzyme-linked immunosorbent assay (icELISA) for the determination of 5 major components of TBA, using a combination of 4 different monoclonal antibodies (MAbs). We prepared 4 MAbs against 5 major bile acids, and determined their cross-reactivity. The coating antigen used in the new method represents a combination of 5 coating antigens (1 : 1 : 1 : 1 : 1 ratio). Inter-assay and intra-assay consistency, and the recovery rate were determined. The standard curve was determined to be linear between 0.2875 and 9.2 μmol L−1, while the IC50 value was 1.85 μmol L−1. Serum TBA concentrations determined using this method were consistent with values obtained by the enzyme cycling method, in both healthy mice and mice with cholestasis, and there is no significant difference between these methods (p > 0.05). We showed that icELISA can be used for the detection of bile acids in human saliva obtained from healthy volunteers, which is not possible with the enzyme cycling method. These results demonstrate that the new icELISA is valid and sensitive enough to determine TBA content in different types of biological samples, and it may be suitable for routine clinical practice.

 Please wait while we load your content...
Please wait while we load your content...