Concise synthesis of calystegines B2 and B3via intramolecular Nozaki–Hiyama–Kishi reaction†
Abstract
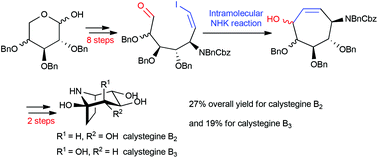
The key step in the concise syntheses of calystegine B2 and its C-2 epimer calystegine B3 was the construction of cycloheptanone 8via an intramolecular Nozaki–Hiyama–Kishi (NHK) reaction of 9, an aldehyde containing a Z-vinyl iodide. Vinyl iodide 9 was obtained by the Stork olefination of aldehyde 10, derived from carbohydrate starting materials. Calystegines B2 (3) and B3 (4) were synthesized from D-xylose and L-arabinose derivatives respectively in 11 steps in excellent overall yields (27% and 19%).
 Please wait while we load your content...
Please wait while we load your content...