Nickel(ii), zinc(ii) and cadmium(ii) complexes of hexapeptides containing separate histidyl and cysteinyl binding sites†
Abstract
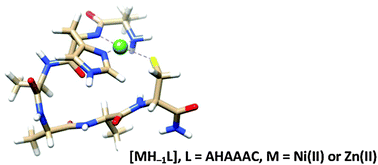
Nickel(II), zinc(II) and cadmium(II) complexes of two N-terminally free hexapeptides AAHAAC and AHAAAC containing separate histidyl and cysteinyl residues have been studied using potentiometric and various spectroscopic (UV-Vis, CD, 1H-NMR) techniques. Both peptides have outstanding metal binding ability but the speciation of the systems and the binding sites of peptides reveal a significant specificity. In the nickel(II)–AAHAAC system the amino terminus is the primary nickel(II) binding site in the form of the (NH2,N−,N−,Nim) chelate. However, the C-terminal thiolate function can bind another nickel(II) ion with the involvement of amide nitrogens in metal binding. Zinc(II) and cadmium(II)ions are not able to promote deprotonation and coordination of the peptide amide groups of AAHAAC and only mononuclear complexes are formed, in which imidazole-N and thiolate-S− donors are the primary metal binding sites. In the case of AHAAAC both nickel(II) and zinc(II) ions can induce deprotonation and coordination of the first amide bond in the sequence. This results in the enhanced stability of the corresponding species, [MH−1L], containing a tridentate (NH2,N−,Nim) binding mode at the amino terminus supported by a macrochelate from the distant thiolate group.

 Please wait while we load your content...
Please wait while we load your content...