Co-catalysis of a bi-functional ligand containing phosphine and Lewis acidic phosphonium for hydroformylation–acetalization of olefins†
Abstract
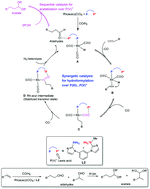
A novel ionic bi-functional ligand of L2 containing a phosphine and a Lewis acidic phosphonium with I− as the counter-anion was prepared and fully characterized. The molecular structure indicated that the bi-functionalities in L2 were well retained without the incompatibility problem for quenching of the acidity of the phosphonium cation by the Lewis basic phosphine fragment or the anionic I− when the incorporated phosphine fragment and the Lewis acidic phosphonium were strictly located in the confined cis-positions. The co-catalysis over L2–Rh(acac)(CO)2 in the ways of synergetic catalysis and sequential catalysis was successfully fulfilled for one-pot hydroformylation–acetalization, which proved not to be the result of the simple mixture of the mono-phosphine (L4) and the phosphonium salt (L4′). In L2, the phosphonium not only acted as a Lewis acid organocatalyst to drive the sequential acetalization of aldehydes, but also contributed to the synergetic catalysis for the preceding hydroformylation through stabilizing the Rh-acyl intermediate with the phosphine cooperatively. The L2-Rh(acac)(CO)2 system is also generally applied to hydroformylation–acetalization of a wide range of olefins in different alcohols. Advantageously, as an ionic phosphonium-based ligand, L2 could be recycled for 7 runs with Rh(acac)(CO)2 together in RTIL of [Bmim]BF4 without obvious activity loss or metal leaching.

 Please wait while we load your content...
Please wait while we load your content...