Catalytic reduction of CO2 by H2 for synthesis of CO, methanol and hydrocarbons: challenges and opportunities
Abstract
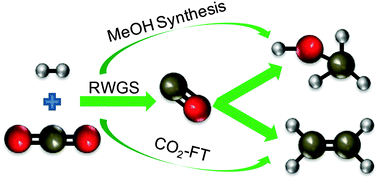
Ocean acidification and climate change are expected to be two of the most difficult scientific challenges of the 21st century. Converting CO2 into valuable chemicals and fuels is one of the most practical routes for reducing CO2 emissions while fossil fuels continue to dominate the energy sector. Reducing CO2 by H2 using heterogeneous catalysis has been studied extensively, but there are still significant challenges in developing active, selective and stable catalysts suitable for large-scale commercialization. The catalytic reduction of CO2 by H2 can lead to the formation of three types of products: CO through the reverse water–gas shift (RWGS) reaction, methanol via selective hydrogenation, and hydrocarbons through combination of CO2 reduction with Fischer–Tropsch (FT) reactions. Investigations into these routes reveal that the stabilization of key reaction intermediates is critically important for controlling catalytic selectivity. Furthermore, viability of these processes is contingent on the development of a CO2-free H2 source on a large enough scale to significantly reduce CO2 emissions.

 Please wait while we load your content...
Please wait while we load your content...