The thermoelectrochemistry of lithium–glyme solvate ionic liquids: towards waste heat harvesting†
Abstract
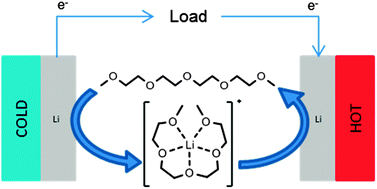
Thermoelectrochemistry offers a simple, scalable technique for direct conversion of waste heat into useful electricity. Here the thermoelectrochemical properties of lithium–glyme solvate ionic liquids, as well as their dilute electrolyte analogues, have been investigated using mixtures of tetraglyme (G4, tetraethylene glycol dimethyl ether) and lithium bis(trifluoromethylsulfonyl)imide (Li[NTf2]). The thermoelectrochemical process is entropically-driven by release of the glyme from the lithium–glyme complex cation, due to electrodeposition of lithium metal at the hotter lithium electrode with concomitant electrodissolution at the cooler lithium electrode. The optimum ratio for thermochemical electricity generation is not the solvate ionic liquid (equimolar mixture of Li[NTf2] and glyme), but rather one Li[NTf2] to four G4, due to the mixtures relatively high ionic conductivity and good apparent Seebeck coefficient (+1.4 mV K−1). Determination of the lithium–glyme mixture thermal conductivity enabled full assessment of the Figure of Merit (ZT), and the efficiency relative to the Carnot efficiency to be determined. As the lithium electrodeposits are porous, alternating the temperature gradient results in a system that actually improves with repeated use.

 Please wait while we load your content...
Please wait while we load your content...