A metal-free synthesis of 2-aminobenzothiazoles through aminyl radical addition to aryl isothiocyanates†
Abstract
A convenient synthesis of 2-aminobenzothiazoles, starting from aryl isothiocyanates and formamides under metal-free conditions, is described. Various secondary and tertiary amine- and even α-amino acid-derived formamides can be used as amino sources in this process. Mechanistic studies suggest that the reaction is initiated by decarbonylative aminyl radical formation in the presence of n-Bu4NI and TBHP, followed by aminyl radical addition to isothiocyanates and cyclization via sulfur centred radical intermediates.
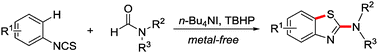

 Please wait while we load your content...
Please wait while we load your content...