Color tuning and energy transfer in Eu2+/Mn2+-doped Ba3Y(PO4)3 eulytite-type orthophosphate phosphors†
Abstract
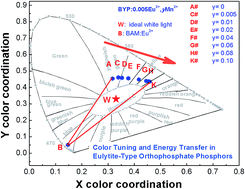
Eulytite-type orthophosphate phosphors Ba3Y(PO4)3:Eu2+, Mn2+ and Ba3Ln(PO4)3:Eu2+ (Ln = Lu, Y and Gd) were synthesized by high-temperature solid state reactions under reductive atmospheres. Their photoluminescence showed a surprising red-shift in the emission spectrum with the increase in the ionic radius of Ln in the Ba3Ln(PO4)3:Eu2+ (Ln = Lu, Y and Gd) phosphors system, which arises from the splitting of the 5d energy level. The phase formation, luminescence properties, and energy-transfer mechanism from the Eu2+ to the Mn2+ ions, and the CIE coordinates in the Ba3Y(PO4)3:Eu2+, Mn2+ phosphors were investigated. From powder X-ray diffraction (XRD) analysis, the formation of the single-phased Ba3Y(PO4)3 in a cubic crystal system with the space group I![[4 with combining macron]](https://www.rsc.org/images/entities/char_0034_0304.gif) 3d (no. 220) was confirmed. With the doping of Mn2+, the spectral overlap between the emission spectrum of Eu2+ and the excitation spectrum of Mn2+ allows resonance-type energy transfer to occur from Eu2+ to Mn2+ with the mechanism carefully studied by luminescence spectra, energy transfer efficiency and decay times. By increasing the Mn2+ doping concentration in the Ba3Y(PO4)3:Eu2+, Mn2+ phosphors, the emission colors can be tuned from yellowish-green through yellow and ultimately to orange. Such color tuning emissions originate from the change in intensity between the 4f–5d transitions of the Eu2+ ions and the 4T1–6A1 transitions of the Mn2+ ions through the energy transfer from the Eu2+ to the Mn2+ ions. In particular, compared with the commercial YAG:Ce phosphor, our developed phosphor contains a larger amount of red-emitting component; thus, it possesses favorable properties for application in warm white LEDs.
3d (no. 220) was confirmed. With the doping of Mn2+, the spectral overlap between the emission spectrum of Eu2+ and the excitation spectrum of Mn2+ allows resonance-type energy transfer to occur from Eu2+ to Mn2+ with the mechanism carefully studied by luminescence spectra, energy transfer efficiency and decay times. By increasing the Mn2+ doping concentration in the Ba3Y(PO4)3:Eu2+, Mn2+ phosphors, the emission colors can be tuned from yellowish-green through yellow and ultimately to orange. Such color tuning emissions originate from the change in intensity between the 4f–5d transitions of the Eu2+ ions and the 4T1–6A1 transitions of the Mn2+ ions through the energy transfer from the Eu2+ to the Mn2+ ions. In particular, compared with the commercial YAG:Ce phosphor, our developed phosphor contains a larger amount of red-emitting component; thus, it possesses favorable properties for application in warm white LEDs.

 Please wait while we load your content...
Please wait while we load your content...