Pd-catalyzed direct C2-acylation and C2,C7-diacylation of indoles: pyrimidine as an easily removable C–H directing group†
Abstract
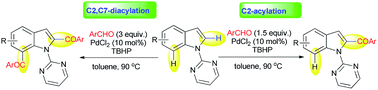
Pyrimidine is successfully used as an easily removable C(sp2)–H directing group for the synthesis of 2-acyl indoles and 2,7-diacyl indoles through direct C–H functionalization using a Pd-catalyst from 1-(pyrimidin-2-yl)-1H-indoles and aldehydes. Easy removal of the pyrimidine directing group using EtONa in DMSO provides C2-acyl indoles.

 Please wait while we load your content...
Please wait while we load your content...