Photoexcited triplet states of UV-B absorbers: ethylhexyl triazone and diethylhexylbutamido triazone†
Abstract
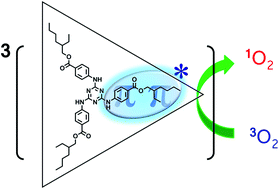
The excited states of UV-B absorbers, ethylhexyl triazone (EHT) and diethylhexylbutamido triazone (DBT), have been studied through measurements of UV absorption, fluorescence, phosphorescence, triplet–triplet absorption and electron paramagnetic resonance spectra in ethanol. The energy levels of the lowest excited singlet (S1) and triplet (T1) states and quantum yields of fluorescence and phosphorescence of EHT and DBT were determined. In ethanol at 77 K, the deactivation process of EHT and DBT is predominantly fluorescence, however, a significant portion of the S1 molecules undergoes intersystem crossing to the T1 state. The observed phosphorescence spectra, T1 lifetimes and zero-field splitting parameters suggest that the T1 state of EHT can be assigned to a locally excited 3ππ* state within p-(N-methylamino)benzoic acid, while the T1 state of DBT can be assigned to a locally excited 3ππ* state within p-(N-methylamino)benzoic acid or p-amino-N-methylbenzamide. The quantum yields of singlet oxygen generation by EHT and DBT were determined by time-resolved near-IR phosphorescence measurements in ethanol at room temperature. EHT and DBT did not exhibit significantly antioxidative properties by quenching singlet oxygen, in contrast to the study by Lhiaubet-Vallet et al.

 Please wait while we load your content...
Please wait while we load your content...