Thermodynamically neutral Kubas-type hydrogen storage using amorphous Cr(iii) alkyl hydride gels†
Abstract
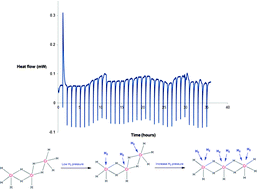
In this paper we present amorphous chromium(III) hydride gels that show promise as reversible room temperature hydrogen storage materials with potential for exploitation in mobile applications. The material uses hydride ligands as a light weight structural feature to link chromium(III) metal centres together which act as binding sites for further dihydrogen molecules via the Kubas interaction, the mode of hydrogen binding confirmed by high pressure Raman spectroscopy. The best material possesses a reversible gravimetric storage of 5.08 wt% at 160 bar and 25 °C while the volumetric density of 78 kgH2 m−3 compares favourably to the DOE ultimate system goal of 70 kg m−3. The enthalpy of hydrogen adsorption is +0.37 kJ mol−1 H2 as measured directly at 40 °C using an isothermal calorimeter coupled directly to a Sieverts gas sorption apparatus. These data support a mechanism confirmed by computations in which the deformation enthalpy required to open up binding sites is almost exactly equal and opposite to the enthalpy of hydrogen binding to the Kubas sites, and suggests that this material can be used in on-board applications without a heat management system.

 Please wait while we load your content...
Please wait while we load your content...