Metal-free oxidative decarbonylative coupling of aromatic aldehydes with arenes: direct access to biaryls†
Abstract
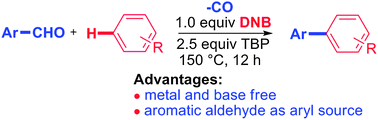
A metal-free oxidative decarbonylative coupling of aromatic aldehydes with electron-rich or electron-deficient arenes to produce biaryl compounds was developed. This novel coupling was proposed to proceed via a non-chain radical homolytic aromatic substitution (HAS) type mechanism, based on the substrate scope, ortho-regioselectivity, radical trapping experiments and DFT calculation studies. With the ready availability of aromatic aldehydes and arenes, metal-free conditions should make this coupling attractive for the biaryl synthesis.

 Please wait while we load your content...
Please wait while we load your content...