A second sphere coordination adduct containing one-dimensional water/methanol channels: X-ray structures, thermal stability and single crystal impedance spectroscopy analysis†‡
Abstract
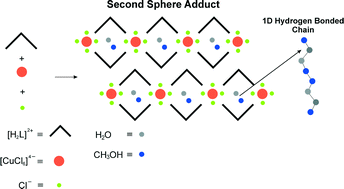
Using the second sphere coordination approach, a new metal complex has been synthesized through self-assembly of a bent shaped organic dication with [CuCl6]4− and Cl− anions, yielding a network with one-dimensional channels. The salt encapsulates water and methanol forming a well-defined hydrogen bonding array. The framework is thermally stable (190 °C) whilst keeping the channels. It can uptake dichloromethane (DCM) but not chloroform from a dichloromethane–chloroform mixture. The solid serves as a support matrix for ionic conductivity via the included water/methanol molecules as shown by single crystal impedance spectroscopy.

 Please wait while we load your content...
Please wait while we load your content...