Freestanding redox buckypaper electrodes from multi-wall carbon nanotubes for bioelectrocatalytic oxygen reduction via mediated electron transfer†
Abstract
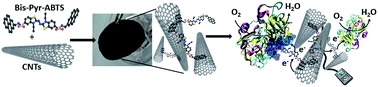
An efficient and easy way of designing free standing redox buckypaper electrodes via the elegant combination of multi-walled carbon nanotubes (MWCNTs) and a bis-pyrene derivative is reported. This bis-pyrene 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid (bis-Pyr-ABTS) acts as a cross-linker between the nanotubes and assures the formation of a mechanically reinforced buckypaper, obtained by a classical filtration technique of a MWCNT suspension in the presence of bis-Pyr-ABTS. In addition, the ABTS derivative assures a mediated electron transfer to laccase. The electroactive buckypapers were characterized in terms of morphology, conductivity, and electrochemical properties. Two setups were evaluated. The first consisted of the immobilization and wiring of laccase enzymes via an inclusion complex formation between the hydrophobic cavity of laccase and the pyrene groups of bis-Pyr-ABTS that are not π-stacked to the nanotubes. The second approach was to evaluate the mediated electron transfer using laccase in solution. For this setup, the developed mediator electrodes demonstrated high performances with maximum currents up to 2 mA ± 70 μA and an excellent operational stability for two weeks with daily one hour discharges using refreshed laccase solutions.

 Please wait while we load your content...
Please wait while we load your content...