Hydriding characteristics of NaMgH2F with preliminary technical and cost evaluation of magnesium-based metal hydride materials for concentrating solar power thermal storage
Abstract
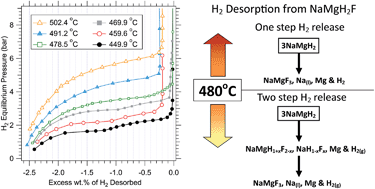
A simplified techno-economic model has been used as a screening tool to explore the factors that have the largest impact on the costs of using metal hydrides for concentrating solar thermal storage. The installed costs of a number of paired metal hydride concentrating solar thermal storage systems were assessed. These comprised of magnesium-based (MgH2, Mg2FeH6, NaMgH3, NaMgH2F) high-temperature metal hydrides (HTMH) for solar thermal storage and Ti1.2Mn1.8H3.0 as the low-temperature metal hydride (LTMH) for hydrogen storage. A factored method approach was used for a 200 MWel power plant operating at a plant capacity factor (PCF) of 50% with 7 hours of thermal storage capacity at full-load. In addition, the hydrogen desorption properties of NaMgH2F have been measured for the first time. It has a practical hydrogen capacity of 2.5 wt% (2.95 wt% theoretical) and desorbs hydrogen in a single-step process above 478 °C and in a two-step process below 478 °C. In both cases the final decomposition products are NaMgF3, Na and Mg. Only the single-step desorption is suitable for concentrating solar thermal storage applications and has an enthalpy of 96.8 kJ mol−1 H2 at the midpoint of the hydrogen desorption plateau. The techno-economic model showed that the cost of the LTMH, Ti1.2Mn1.8H3.0, is the most significant component of the system and that its cost can be reduced by increasing the operating temperature and enthalpy of hydrogen absorption in the HTMH that, in turn, reduces the quantity of hydrogen required in the system for an equivalent electrical output. The result is that, despite the fact that the theoretical thermal storage capacity of NaMgH2F (1416 kJ kg−1) is substantially lower than the theoretical values for MgH2 (2814 kJ kg−1), Mg2FeH6 (2090 kJ kg−1) and NaMgH3 (1721 kJ kg−1), its higher enthalpy and operating temperature leads to the lowest installed cost of the systems considered. A further decrease in cost could be achieved by utilizing metal hydrides with yet higher enthalpies and operating temperatures or by finding a lower cost option for the LTMH.

 Please wait while we load your content...
Please wait while we load your content...