Brønsted acid promoted addition–cyclization and C–C bond cleavage: a convenient synthesis of 2-amino-5-aroylmethylthiazoles derivatives†
Abstract
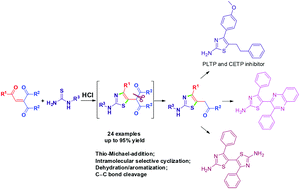
A Brønsted acid promoted C–C bond cleavage method for the synthesis of novel 2-amino-5-aroylmethylthiazole derivatives has been directly developed from 1,4-enediones and thioureas through self-sequenced thio-Michael-addition, intramolecular selective cyclization, dehydration/aromatization, and C–C bond cleavage reactions. It is noteworthy that this reaction has significant advantages in simple reagents, under environmentally benign conditions and with excellent yields. This highly efficient method is also a highly attractive alternative for the preparation of PLTP, CETP inhibitors and novel biheterocycles.

 Please wait while we load your content...
Please wait while we load your content...