Pyridine-phosphinimine ligand-accelerated Cu(i)-catalyzed azide–alkyne cycloaddition for preparation of 1-(pyridin-2-yl)-1,2,3-triazole derivatives†
Abstract
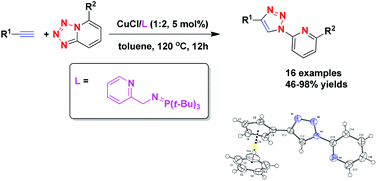
A series of phosphinimine ligands were designed and used in the Cu(I)-catalyzed azide–alkyne cycloaddition (CuAAC) reaction of tetrazolo[1,5-a]pyridines and alkynes for the first time. By optimizing the reaction conditions, an efficient catalytic system (CuCl/2-PyCH2N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) PtBu3) was developed to give 1-(pyridin-2-yl)-1,2,3-triazole derivatives in moderate to excellent yields (46–98%).
PtBu3) was developed to give 1-(pyridin-2-yl)-1,2,3-triazole derivatives in moderate to excellent yields (46–98%).

 Please wait while we load your content...
Please wait while we load your content...