Lead identification and structure–activity relationships of heteroarylpyrazole arylsulfonamides as allosteric CC-chemokine receptor 4 (CCR4) antagonists†
Abstract
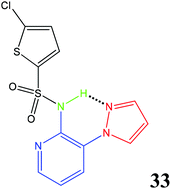
A knowledge-based library of aryl 2,3-dichlorophenylsulfonamides was synthesised and screened as human CCR4 antagonists, in order to identify a suitable hit for the start of a lead-optimisation programme. X-ray diffraction studies were used to identify the pyrazole ring as a moiety that could bring about intramolecular hydrogen bonding with the sulfonamide NH and provide a clip or orthogonal conformation that was believed to be the preferred active conformation. Replacement of the core phenyl ring with a pyridine, and replacement of the 2,3-dichlorobenzenesulfonamide with 5-chlorothiophenesulfonamide provided compound 33 which has excellent physicochemical properties and represents a good starting point for a lead optimisation programme. Electronic structure calculations indicated that the preference for the clip or orthogonal conformation found in the small molecule crystal structures of 7 and 14 was in agreement with the order of potency in the biological assay.

 Please wait while we load your content...
Please wait while we load your content...