Synthesis and biological evaluation of coumarin–1,2,3-triazole–dithiocarbamate hybrids as potent LSD1 inhibitors†
Abstract
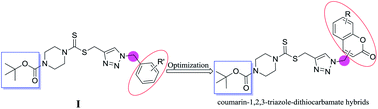
Two series of coumarin–1,2,3-triazole–dithiocarbamate hybrids were designed, synthesized and evaluated for their inhibitory activity towards lysine specific demethylase 1 (LSD1). Compounds 8a, 8d–8f, 8i–8l presented potent activity against lysine specific demethylase 1. Among them, compound 8k showed potent and reversible inhibition against lysine specific demethylase 1 with an IC50 value of 0.39 μM, which was 74-fold more potent than that of tranylcypromine (2-PCPA). Besides, compound 8k displayed excellent selectivity against lysine specific demethylase 1 without inhibition against monoamine oxidases (MAOs) A and B. Further investigation revealed that compound 8k was active at both recombinant and cell levels by upregulating the expression of H3K4me1, H3K4me2 and H3K9me2.

 Please wait while we load your content...
Please wait while we load your content...