Mixed amido-/imido-/guanidinato niobium complexes: synthesis and the effect of ligands on insertion reactions†‡
Abstract
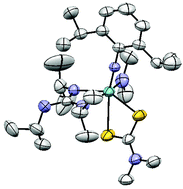
The new monoguanidinato complexes [Nb(NMe2)2{N(2,6-iPr2C6H3)}{(NR)(NR′)C(NMe2)}] (R = R′ = iPr, 2; R = tBu, R′ = Et, 3) were obtained by the insertion reaction of either diisopropylcarbodiimide or 1-tert-butyl-3-ethylcarbodiimide with the triamido precursor [Nb(NMe2)3(N-2,6-iPr2C6H3)] (1) bearing a bulky imido moiety. The μ-oxo derivative [{N(2,6-iPr2C6H3)}{(NiPr)2C(NMe2)}(NMe2)Nb]2(μ-O) (2a) was formed by an unexpected hydrolysis reaction of the amido niobium compound 2. Alternatively, monoguanidinato complexes [Nb(NMe2)2{N(2,6-iPr2C6H3)}{(NiPr)2C(NHR)}] (R = iPr, 4, nBu, 5) can be obtained by protonolysis of 1 with N,N′,N′′-alkylguanidines [(NHiPr)2C(NR)] (R = iPr, nBu). Compound 1 also reacts with either tert-butylisocyanide or 2,6-xylylisocyanide to give, by a migratory insertion reaction, the corresponding iminocarbamoyl compounds [Nb(NMe2)2{(NMe2)C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NR}{N(2,6-iPr2C6H3)}] (R = tBu, 6, Xy, 7). Addition of the neutral alkylguanidines to complex 6 results in a facile C–N bond cleavage at room temperature in a process directed by the formation of the stable chelate complex 4 or 5. Complex 1 reacts with heterocumulenic CS2 to produce new imido dithiocarbamato complexes [Nb(NMe2){S2C(NMe2)}2{N(2,6-iPr2C6H3)}] (8) and [Nb{S2C(NMe2)}3{N(2,6-iPr2C6H3)}] (9). These complexes do not react with alkylguanines, although new mixed guanidinato dithiocarbamato complexes [Nb(NMe2){S2C(NMe2)}{(NiPr)2C(NHiPr)}{N(2,6-iPr2C6H3)}] (10) and [Nb{(S2C(NMe2)}2{(NiPr)2C(NHiPr)}{N(2,6-iPr2C6H3)}] (11) can be obtained by reaction of complex 4 with one or two equivalents of CS2, respectively. All of the complexes were characterized spectroscopically and the dynamic behaviour of some of them was studied by variable-temperature NMR. The molecular structures of 2a, 3, 6 and 10 were also established by X-ray diffraction studies.
NR}{N(2,6-iPr2C6H3)}] (R = tBu, 6, Xy, 7). Addition of the neutral alkylguanidines to complex 6 results in a facile C–N bond cleavage at room temperature in a process directed by the formation of the stable chelate complex 4 or 5. Complex 1 reacts with heterocumulenic CS2 to produce new imido dithiocarbamato complexes [Nb(NMe2){S2C(NMe2)}2{N(2,6-iPr2C6H3)}] (8) and [Nb{S2C(NMe2)}3{N(2,6-iPr2C6H3)}] (9). These complexes do not react with alkylguanines, although new mixed guanidinato dithiocarbamato complexes [Nb(NMe2){S2C(NMe2)}{(NiPr)2C(NHiPr)}{N(2,6-iPr2C6H3)}] (10) and [Nb{(S2C(NMe2)}2{(NiPr)2C(NHiPr)}{N(2,6-iPr2C6H3)}] (11) can be obtained by reaction of complex 4 with one or two equivalents of CS2, respectively. All of the complexes were characterized spectroscopically and the dynamic behaviour of some of them was studied by variable-temperature NMR. The molecular structures of 2a, 3, 6 and 10 were also established by X-ray diffraction studies.

 Please wait while we load your content...
Please wait while we load your content...