Synthesis, structure and spectral and electrochemical properties of 3-pyrrolyl BODIPY-metal dipyrrin complexes†
Abstract
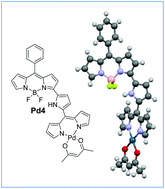
The stable dipyrrin substituted 3-pyrrolyl BODIPY (α-dipyrrin 3-pyrrolyl BODIPY) was synthesized by oxidation of dipyrromethane substituted 3-pyrrolyl BODIPY with 2,3-dichloro-5,6-dicyano-benzoquinone in CH2Cl2 at room temperature. The α-dipyrrin 3-pyrrolyl BODIPY is characterized by using HR-MS, 1D, 2D NMR and absorption spectroscopic techniques. The absorption spectrum of α-dipyrrin 3-pyrrolyl BODIPY showed a characteristic absorption band at 630 nm and a charge transfer band at 717 nm due to intramolecular charge transfer from the dipyrrin unit to the 3-pyrrolyl BODIPY unit. The 3-pyrrolyl BODIPY-metal dipyrrin complexes (Pd(II), Re(I) and Ru(II)) were prepared by treating α-dipyrrin 3-pyrrolyl BODIPY with appropriate metal salts in toluene–triethylamine at 100 °C and purified by silica gel column chromatography. The crystal structure obtained for the 3-pyrrolyl BODIPY-Pd(II) dipyrrin complex showed that the 3-pyrrolyl BODIPY and metal dipyrrin moieties are aligned to each other with an angle of 41.9°. The absorption studies showed a strong band at ∼620 nm corresponding to 3-pyrrolyl BODIPY moiety and a weak band at ∼530 nm corresponding to metal dipyrrin unit with complete disappearance of the charge transfer band at 717 nm. The complexes are electron deficient and exhibited only reversible/quasi-reversible reductions in cyclic voltammetry.

 Please wait while we load your content...
Please wait while we load your content...