Inter-layer charge disproportionation in the dual-layer organic metal (tTTF-I)2ClO4 with unsymmetrical I⋯O halogen bond interactions†
Abstract
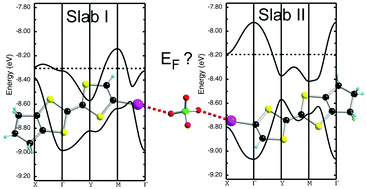
A mixed-valence salt of tTTF-I with ClO4−, formulated as (tTTF-I)2ClO4, is characterized by the presence of two crystallographically independent donor molecules, segregated in different layers and linked together through I⋯O interactions with the ClO4− anion disordered at room temperature. The tTTF-I donor molecule was prepared by metallation of tTTF (trimethylene tetrathiafulvalene) followed by reaction with iodine to afford the mono and diiodo derivatives tTTF-I and tTTFI2, respectively. The crystal structure of the latter neutral tTTFI2 shows the occurrence of strong type II, I⋯I halogen bond interactions. Band structure calculations of the dual-layer structure of the 2 : 1 salt (tTTF-I)2ClO4 show co-existence of both 1D open and 2D closed Fermi surfaces. The salt undergoes a metal–insulator phase transition at TMI = 90 K, associated with an electronic dimensionality decrease, since already at 100 K, the 2D part of the Fermi surface transforms into 1D corrugated planes. High resolution X-ray investigations performed at 100 K, combined with multipolar refinements, indicate an approximately equivalent +0.5e charge in both donor molecules, as also deduced from the intramolecular bond distances. On the other hand, Raman spectroscopic investigations show that at ambient temperature the charge is actually distributed non-uniformly in conducting layers of tTTF-I molecules, with the identification of molecules with charges +1, +0.5, 0e, while at low temperature the charge distribution becomes essentially uniform (+0.5e), as confirmed from the X-ray high resolution data. These apparently contradictory behaviors are actually a consequence of a partial electron transfer between the two independent slabs to reach a common Fermi level in the metallic phase.

 Please wait while we load your content...
Please wait while we load your content...