Coulomb repulsion versus cycloaddition: formation of anionic four-membered rings from sodium phosphaethynolate, Na(OCP)†
Abstract
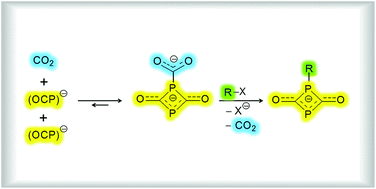
Carbon dioxide and two equivalents of Na(OCP) form, in an equilibrium reaction, a CO2 adduct of the composition Na2(P2C3O4). The anion of this salt, [O2C–P(CO)2P]2−, is built up by a four-membered 1,3-diphosphetane-2,4-dione ring and a carboxylate unit attached to one of the phosphorus atoms. A remarkable π-delocalization was observed within the OCPCO moiety. The stepwise reaction mechanism leading to Na2(P2C3O4) was investigated with quantum chemical calculations. Accompanied by the release of CO2, Na2(P2C3O4) reacts with both 2-iodopropane and 4,4′,4′′-trimethoxytriphenylmethyl chloride to form four-membered cyclic anions. For comparison the analogous reactions were performed with Na(OCP) instead of Na2(P2C3O4) and the results are discussed in detail.

 Please wait while we load your content...
Please wait while we load your content...