Ni(ii) binding to the 429–460 peptide fragment from human Toll like receptor (hTLR4): a crucial role for nickel-induced contact allergy?†
Abstract
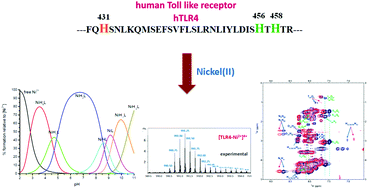
The FQH431SNLKQMSEFSVFLSLRNLIYLDISH456TH458TR fragment, containing three histidine residues, the conserved H431 and the non-conserved H456 and H458, located from 429 to 460 amino acid residues in the C-terminal portion of human Toll-like-receptor 4 (hTLR4), which is directly activated by nickel, a well known contact allergen, has been tested for Ni(II) binding. The complex formation capability of the 32-amino acid sequence with Ni(II) ions has been followed by potentiometric, UV-Vis, CD, MS and NMR measurements. Ni(II) is able to bind to all three histidines by forming macrocycle complexes at low and physiological pH. From pH 9 on, a 4N diamagnetic species (Nim, 3Nam−) with the participation of an imidazole nitrogen and three deprotonated nitrogens from His28, Ser27 and Ile26 amides from the backbone of the model peptide has been determined. From the NMR results it was possible to determine that His28, which mimics the H456 residue in the protein, together with the environment around it, was mainly involved in the binding.

 Please wait while we load your content...
Please wait while we load your content...