Glycol metalloporphyrin derivatives in solution or immobilized on LDH and silica: synthesis, characterization and catalytic features in oxidation reactions†
Abstract
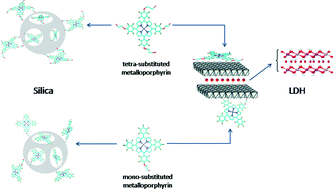
We reacted [meso-tetrakis(pentafluorophenyl)porphyrin] with ethylene glycol to obtain porphyrins substituted with one to four hydroxyalkyloxy groups at position 4 of the meso-aryl groups. We then inserted iron(III) or manganese(III) into the mono- and tetra-substituted free-base derivatives, which afforded the corresponding metalloporphyrin complexes. Next, we immobilized the iron(III) and manganese(III) porphyrins on two supports: layered double hydroxide (LDH) and silica (synthesized by the sol–gel process). We characterized the resulting solids using powder X-ray diffraction (PXRD), UV–vis electronic spectroscopy, and electron paramagnetic resonance (EPR), and we investigated the catalytic activity of the materials in both homogeneous and heterogeneous media. The metalloporphyrins provided good catalytic conversions in (Z)-cyclooctene oxidation. As for cyclohexane oxidation, the catalysts were selective for the alcohol instead of the ketone. In the case of heterogeneous catalysis, the immobilized metalloporphyrins furnished slightly lower yields as compared with homogeneous catalysis. However, these solids presented a major advantage: reusability. Indeed, these solid catalysts retained their activity for at least three cycles in the case of (Z)-cyclooctene oxidation.

 Please wait while we load your content...
Please wait while we load your content...