The alternation effect in ionic liquid homologous series
Abstract
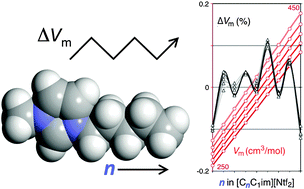
A recent report on a density odd–even alternation effect in a homologous series of ionic liquids (alkyltrioctylphosphonium chlorides, with the linear alkyl group ranging from ethyl to decyl) led to the detection of a similar trend in another ionic liquid family based on a different cation (1-alkyl-3-methylimidazolium). Ab initio calculations and Molecular Dynamics simulations of the corresponding ions confirmed that the charge distribution along the alkyl side chains and the conformations adopted by them are not the direct cause of the odd–even effect. The simulations also showed that all cation side chains tend to adopt transoid conformations that pack head-to-head in the liquid phase. Such types of conformations/packing lead to odd–even alternation effects on properties involving solid phases of different molecular compounds containing linear alkyl chains. The surprising results obtained for the ionic liquid series enabled us to unveil similar trends in the liquid phases of linear alkanes and alkanols via the application of a simple corresponding states principle.

 Please wait while we load your content...
Please wait while we load your content...