Highest measured anodic stability in aqueous solutions: graphenic electrodes from the thermolyzed asphalt reaction†
Abstract
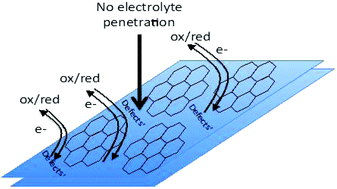
Graphenic material synthesized by the University of Idaho Thermolyzed Asphalt Reaction (GUITAR) was characterized by cyclic voltammetry. Despite morphological similarities, GUITAR maintains distinct characteristics as an electrode material versus graphene, graphite, pyrolytic graphite and highly ordered pyrolytic graphite (HOPG). Specifically, GUITAR possesses a combination of (i) facile electron transfer with redox species (ii) very high anodic corrosion potentials. Cyclic voltammetric ΔEp studies of Ru(NH3)63+/2+ (71 mV) and Fe(CN)63−/4− (80 mV) in 1 M KCl (aq) indicate facile electron transfer kinetics. This is unlike HOPG and graphene electrodes which have a barrier to heterogeneous electron transfer. The anodic limit of GUITAR is 2.7 V vs. SHE at 200 μA cm−2 in 1 M H2SO4 and demonstrates anodic stability greater than boron doped diamond electrodes. In this characteristic, GUITAR exceeds the corrosion potential of all other sp2-hydribized carbon materials by 700 mV in 1 M H2SO4. Hypotheses on the origins for these distinctive characteristics along with preliminary studies of GUITAR's performance as a dimensionally stable anode with methylene blue electro-oxidation are presented.

 Please wait while we load your content...
Please wait while we load your content...