Examination of the chemical behavior of the quercetin radical cation towards some bases†
Abstract
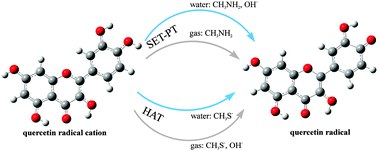
It has been generally accepted that, due to high ionization potential values, single
* Corresponding authors
a
Department of Chemical-Technological Sciences, State University of Novi Pazar, Vuka Karadžića bb, 36300 Novi Pazar, Serbia
E-mail:
zmarkovic@np.ac.rs
b
Faculty of Agriculture, The Josip Juraj Strossmayer University, HR-31107 Osijek, Croatia
E-mail:
damic@pfos.hr
c
Bioengineering Research and Development Center, 6 Prvoslava Stojanovića, 34000 Kragujevac, Serbia
E-mail:
deki82@kg.ac.rs
d
Faculty of Physical Chemistry, University of Belgrade, 12-16 Studentski trg, 11000 Belgrade, Serbia
E-mail:
markovich@ffh.bg.ac.rs
e
Faculty of Science, University of Kragujevac, 12 Radoja Domanovića, 34000 Kragujevac, Serbia
E-mail:
mark@kg.ac.rs
It has been generally accepted that, due to high ionization potential values, single

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
Z. Marković, D. Amić, D. Milenković, J. M. Dimitrić-Marković and S. Marković, Phys. Chem. Chem. Phys., 2013, 15, 7370 DOI: 10.1039/C3CP44605K
To request permission to reproduce material from this article, please go to the Copyright Clearance Center request page.
If you are an author contributing to an RSC publication, you do not need to request permission provided correct acknowledgement is given.
If you are the author of this article, you do not need to request permission to reproduce figures and diagrams provided correct acknowledgement is given. If you want to reproduce the whole article in a third-party publication (excluding your thesis/dissertation for which permission is not required) please go to the Copyright Clearance Center request page.
Read more about how to correctly acknowledge RSC content.
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
