Thermodynamic considerations for solubility and conformational transitions of poly-N-isopropyl-acrylamide†
Abstract
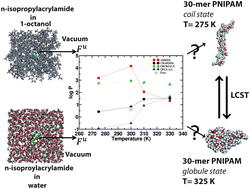
Thermodynamic considerations based on the free energy of hydration, free energy of solvation and partition coefficient predictions of the monomer N-isopropyl-acrylamide (NIPAM) determined using various intermolecular potentials are used to elucidate the origin of hydrophobicity/hydrophilicity across the lower critical solution temperature (LCST). Thermodynamic properties are predicted for NIPAM using adaptive bias force-molecular dynamics and various popular force-fields (AMBER, OPLS-AA, CHARMM and GROMOS) at four different temperatures: below the LCST (275 K and 300 K) and above the LCST (310 K and 330 K). The effect of changes in the thermodynamic properties of the monomer NIPAM at various temperatures below and above LCST on the kinetics of conformational transition of thermo-sensitive

 Please wait while we load your content...
Please wait while we load your content...