Site-directed antibody immobilization using a protein A–gold binding domain fusion protein for enhanced SPR immunosensing
Abstract
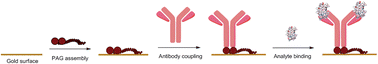
We have implemented a novel strategy for the oriented immobilization of antibodies onto a gold surface based on the use of a fusion

 Please wait while we load your content...
Please wait while we load your content...