Mass spectrometry methods for intrinsically disordered proteins
Abstract
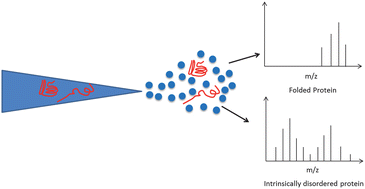
In the last ten years mass spectrometry has emerged as a powerful biophysical technique capable of providing unique insights into the structure and dynamics of proteins. Part of this explosion in use involves investigations of the most recently ‘discovered’ subset of proteins: the so-called ‘Intrinsically Disordered’ or ‘Natively Unstructured’ proteins. A key advantage of the use of mass spectrometry to study intrinsically disordered proteins (IDPs) is its ability to test biophysical assertions made about why they differ from structured proteins. For example, from the charge state distribution presented by a protein following nano-electrospray (n-ESI) it is possible to infer the range of conformations present in solution and hence the extent of disorder; n-ESI is highly sensitive to the degree of folding at the moment of transfer from the liquid to the gas phase. The combination of mass spectrometry with ion mobility (IM-MS) provides rotationally averaged collision cross-sections of molecular ions which can be correlated with conformation; this too can be applied to IDPs. Another feature which can be monitored by IM-MS is the tendency of disordered proteins to form amyloid fibrils, the protein aggregates involved in the onset of neurodegenerative diseases such as Parkinson's and Alzheimer's. IM-MS provides a useful insight into events that occur during the early stages of aggregation including delineating the structure of the monomer, identifying oligomer distributions, and revealing mechanistic details of the aggregation process. Here we will review the use of MS and IM-MS to study IDPs using examples from our own and other laboratories.

 Please wait while we load your content...
Please wait while we load your content...