Activation of aliphatic C–H bonds by tetracyanobenzene photosensitization. A time-resolved and steady-state investigation†
Abstract
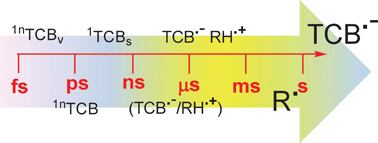
The photochemistry of
* Corresponding authors
a
PhotoGreen Lab, Department of Chemistry, University of Pavia, V.le Taramelli 12, Pavia, Italy
E-mail:
angelo.albini@unipv.it
Fax: (+)390382987323
Tel: (+)390382987316
b Istituto per la Sintesi Organica e la Fotoreattività (ISOF-CNR), Via P. Gobetti 101, Bologna, Italy
c Physikalisch-Chemisches Institut, Universität Zürich, Winterthurerstrasse, Zürich, Switzerland
d Laboratoire de Spectrochimie Infrarouge et Raman-LASIR, (UMR 8516 du CNRS), Université des Sciences et Technologies de Lille, Bat C5, Villeneuve d'Ascq Cedex, France
The photochemistry of

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
S. Protti, M. Fagnoni, S. Monti, J. Réhault, O. Poizat and A. Albini, RSC Adv., 2012, 2, 1897 DOI: 10.1039/C2RA01054B
To request permission to reproduce material from this article, please go to the Copyright Clearance Center request page.
If you are an author contributing to an RSC publication, you do not need to request permission provided correct acknowledgement is given.
If you are the author of this article, you do not need to request permission to reproduce figures and diagrams provided correct acknowledgement is given. If you want to reproduce the whole article in a third-party publication (excluding your thesis/dissertation for which permission is not required) please go to the Copyright Clearance Center request page.
Read more about how to correctly acknowledge RSC content.
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
