A valence bond view of isocyanides' electronic structure†
Abstract
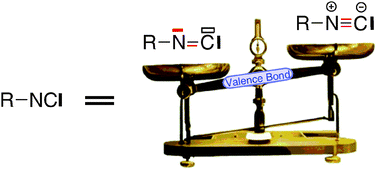
High level Valence Bond calculations support a predominantly carbenic electronic structure for
* Corresponding authors
a
Ecole Normale Supérieure de Lyon, 46, alléed'Italie, F-69364 Lyon Cedex 07, France
E-mail:
paul.fleurat-lessard@ens-lyon.fr
Fax: +33 4 72 72 88 60
b Laboratoire Chimie et Procédés, UMR 7652 CNRS-DCSO-ENSTA, École Nationale Supérieure de Techniques Avancées, 32 boulevard Victor, F-75739 Paris Cedex 15, France
c
Laboratoire de Chimie Théorique, UMR 7616, UPMC Univ Paris 06, case courrier 137, 4 place Jussieu, F-75252 Paris Cedex 05, France
E-mail:
benoit.braida@upmc.fr
Fax: +33 4 72 72 88 60
d Laboratoire de Chimie Physique, CNRS UMR 8000, Université de Paris-Sud, F-91405 Orsay, France
High level Valence Bond calculations support a predominantly carbenic electronic structure for

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
R. Ramozzi, N. Chéron, B. Braïda, P. C. Hiberty and P. Fleurat-Lessard, New J. Chem., 2012, 36, 1137 DOI: 10.1039/C2NJ40050B
To request permission to reproduce material from this article, please go to the Copyright Clearance Center request page.
If you are an author contributing to an RSC publication, you do not need to request permission provided correct acknowledgement is given.
If you are the author of this article, you do not need to request permission to reproduce figures and diagrams provided correct acknowledgement is given. If you want to reproduce the whole article in a third-party publication (excluding your thesis/dissertation for which permission is not required) please go to the Copyright Clearance Center request page.
Read more about how to correctly acknowledge RSC content.
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
