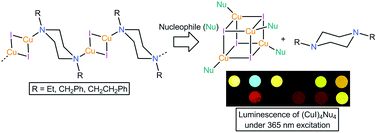
The synthesis, X-ray structures and photophysics of ten complexes of CuX (X = I or Br) with bridging N-substituted and N,N′-disubstituted piperazines (Pip) are presented. Depending on the steric demand of the Pip substituents, the complexes fall into four categories: (CuX)4(Pip)2, which are networks of linked Cu4X4 cubane units, (CuX)2(Pip), which are chains of linked Cu2X2 rhombs, and (CuX)2(Pip)2 or (CuX)4(Pip)4, which are simple rhomboid dimers and cubane tetramers. A combination of spectroscopic studies and DFT calculations was used to investigate the luminescence of the products. The results suggest that the relatively high energy emission seen in dimers is due to cluster-centred (XMLT/metal-centred) excitations for the aliphatic amines and MLCT (d → π*) for aromatic amines, and low energy emission seen in the tetramers is the result of cluster-centred transitions. The (CuI)2(Pip) complexes act as sensor materials, undergoing irreversible reaction with aliphatic and aromatic amines (Nu) in the vapour state, irreversibly producing cubanes (CuI)4Nu4, with corresponding production of long wavelength emission.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...