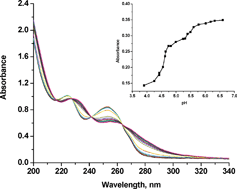
The novel dinuclear Pt(II) complexes [{trans-Pt(NH3)2Cl}2(μ-pyrazine)](ClO4)2 (Pt1), [{trans-Pt(NH3)2Cl}2(μ-4,4′-bipyridyl)](ClO4)2·DMF (Pt2), and [{trans-Pt(NH3)2Cl}2(μ-1,2-bis(4-pyridyl)ethane)](ClO4)2 (Pt3), were synthesized. Acid–base titrations, and temperature and concentration dependent kinetic measurements of the reactions with biologically relevant ligands such as thiourea (Tu), glutathione (GSH) and guanosine-5′-monophosphate (5′-GMP) were studied at pH 2.5 and 7.2. The reactions were followed under pseudo-first-order conditions by stopped-flow and UV-vis spectrophotometry. 1H NMR spectroscopy was used to follow the substitution of chloride in the complex [{trans-Pt(NH3)2Cl}2(μ-4,4′-bipyridyl)](ClO4)2·DMF by guanosine-5′-monophosphate (5′-GMP) under second-order conditions. The results indicate that the bridging ligand has an influence on the reactivity of the complexes towards nucleophiles. The order of reactivity of the investigated complexes is Pt1 > Pt2 > Pt3.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...