Charge regulation in redox active monolayers embedded in proton exchanger surfaces
Abstract
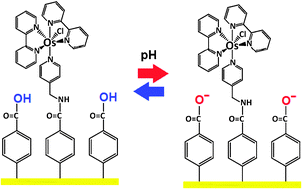
Experimental evidence of the variation in redox potential of osmium pyridine complexes tethered to a Au surface by thiol or diazonium chemistry at different ionic concentration and electrolyte pH is described. The interplay between the charged redox and acid groups is described by a charge regulation model based on PM-IRRAS spectroscopic evidence on the different degrees of surface protonation.

 Please wait while we load your content...
Please wait while we load your content...