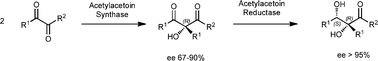
An enzymatic strategy for the preparation of optically pure α-alkyl-α,β-dihydroxyketones is reported. Homo- and cross-coupling reactions of α-diketones catalyzed by acetylacetoin synthase (AAS) produce a set of α-alkyl-α-hydroxy-β-diketones (30–60%, ee 67–90%), which in turn are reduced regio-, diastereo-, and enantioselectively to the corresponding chiral α-alkyl-α,β-dihydroxyketones (60–70%, ee >95%) using acetylacetoin reductase (AAR) as catalyst. Both enzymes are obtained from Bacillus licheniformis and used in a crude form. The relative syn stereochemistry of the enantiopure α,β-dihydroxy products is assigned by NOE experiments, whereas their absolute configuration is determined by conversion of the selected 3,4-dihydroxy-3-methyl-pentan-2-one to the natural product (+)-citreodiol.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...