The phenanthridinebiguanides efficiently differentiate between dGdC, dAdT and rArU sequences by two independent, sensitive spectroscopic methods†
Abstract
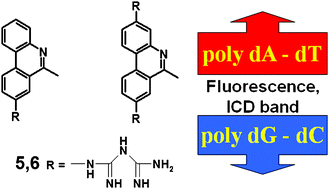
At submicromolar concentrations two novel
* Corresponding authors
a
Division of Organic Chemistry and Biochemistry, Ruđer Bošković Institute, Bijenička cesta 54, PO Box 180, HR-10002 Zagreb, Croatia
E-mail:
pianta@irb.hr
b Laboratory of Analytical Chemistry, Department of Chemistry, Faculty of Science, University of Zagreb, Horvatovac 102a, HR-10000 Zagreb, Croatia
c Department of Medicinal Chemistry and Biochemistry, School of Medicine, J.J. Strossmayer University of Osijek, 31000 Osijek, Croatia
d Clinical Institute of Nuclear Medicine and Radiation Protection, University Hospital Centre Osijek, 31000 Osijek, Croatia
At submicromolar concentrations two novel

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
M. Radić Stojković, S. Miljanić, K. Mišković, L. Glavaš-Obrovac and I. Piantanida, Mol. BioSyst., 2011, 7, 1753 DOI: 10.1039/C1MB05030C
To request permission to reproduce material from this article, please go to the Copyright Clearance Center request page.
If you are an author contributing to an RSC publication, you do not need to request permission provided correct acknowledgement is given.
If you are the author of this article, you do not need to request permission to reproduce figures and diagrams provided correct acknowledgement is given. If you want to reproduce the whole article in a third-party publication (excluding your thesis/dissertation for which permission is not required) please go to the Copyright Clearance Center request page.
Read more about how to correctly acknowledge RSC content.
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
