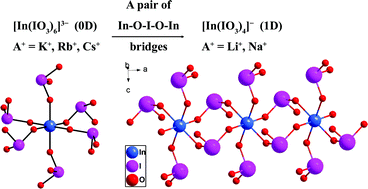
Systematic explorations of new phases in the AI–InIII–IV–O system by hydrothermal reactions led to five new compounds, namely, AIn(IO3)4 (A = Li, Na), Rb3In(IO3)6 and A2HIn(IO3)6 (A = Rb, Cs). The structure of AIn(IO3)4 (A = Li, Na) contains one-dimensional [In(IO3)4]− chains separated by Li+ or Na+ cations. In both compounds, each In3+ cation is octahedrally coordinated by six IO3− anions, neighboring In3+ cations are interconnected by bidentate bridging iodate anions into 1D chains. The structures of Rb3In(IO3)6 and A2HIn(IO3)6 (A = Rb, Cs) all feature isolated [In(IO3)6]3− anions with alkali metal ions (and H+ ions) as spacers. Both optical diffuse reflectance spectrum measurements and band structure calculations based on DFT methods indicate that LiIn(IO3)4, NaIn(IO3)4, and Rb2HIn(IO3)6 are insulators.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...