Monitoring selected hydrogen bonds in crystal hydrates of amino acid salts: combining variable-temperature single-crystal X-ray diffraction and polarized Raman spectroscopy†
Abstract
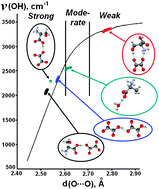
Predicting behaviour of hydrogen bonds with varying temperature, in particular-correlating donor–acceptor distances in the O–H⋯O hydrogen bonds with the frequencies of O–H stretching vibrations is important for understanding dynamics of biomolecules and phase transitions in crystals. A commonly used correlation suggested earlier in the literature is based on statistical analysis of different compounds [A. Novak, Structure and Bonding, 1974, 18, 177; K. Nakamoto, M. Margoshes, R. E. Rundle, J. Am. Chem. Soc., 1955, 77, 6480]. The present study is a rare example when correlations between geometry and energy parameters have been found for selected individual hydrogen bonds in the same crystalline compound at multiple temperatures. The properties of several types of O–H⋯O hydrogen bonds in

 Please wait while we load your content...
Please wait while we load your content...