Oscillations death revisited; coupling of identical chemical oscillators
Abstract
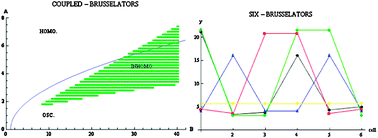
The coupling of identical reactors containing chemical oscillators is discussed. The coupling is executed by means of transferring chemical species from one cell (reactor) to the other in a diffusion like manner i.e. in proportion to the concentration difference between the cells. The coupling rate constant, however, is the same for all species. The individual, uncoupled, cells may be oscillating or in a stable steady state (the same for all reactors). In both cases, depending on the initial conditions, the symmetry breaks, and the cells may end up—contrary to intuition—in a stable steady state in which the final concentrations are not equal in the various reactors. The Brusselator and Oregonator mechanisms are examined and they behave in the manner described. On the other hand, the Lotka-Volterra mechanism, being conservative, keeps, when coupled, only the homogeneous solutions.

 Please wait while we load your content...
Please wait while we load your content...