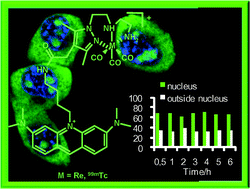
Tricarbonyl M(I) (M = Re, 99mTc) complexes bearing acridine fluorophores: synthesis, characterization, DNA interaction studies and nuclear targeting†
Abstract
New
* Corresponding authors
a
Unidade de Ciências Químicas e Radiofarmacêuticas, ITN, Estrada Nacional 10, 2686-953 Sacavém, Portugal
E-mail:
isantos@itn.pt
b Centro de Química Estrutural, IST, TU Lisbon, Av.Rovisco Pais, 1049-001 Lisboa, Portugal
c IMM, Faculdade de Medicina da Universidade de Lisboa, Av. Prof. Egas Moniz, 1649-028 Lisboa, Portugal
d Department of Pharmaceutical Sciences, University of Padova, via Marzolo 5, 35131 Padova, Italy
New

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
T. Esteves, C. Xavier, S. Gama, F. Mendes, P. D. Raposinho, F. Marques, A. Paulo, J. C. Pessoa, J. Rino, G. Viola and I. Santos, Org. Biomol. Chem., 2010, 8, 4104 DOI: 10.1039/C0OB00073F
To request permission to reproduce material from this article, please go to the Copyright Clearance Center request page.
If you are an author contributing to an RSC publication, you do not need to request permission provided correct acknowledgement is given.
If you are the author of this article, you do not need to request permission to reproduce figures and diagrams provided correct acknowledgement is given. If you want to reproduce the whole article in a third-party publication (excluding your thesis/dissertation for which permission is not required) please go to the Copyright Clearance Center request page.
Read more about how to correctly acknowledge RSC content.
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
