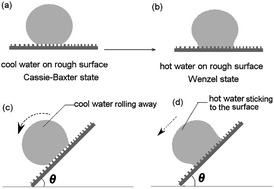
Superhydrophobic surfaces that can repel hot liquids stand for a new generation of self-cleaning surfaces with broad applications. However, can current existing superhydrophobic surfaces repel hot water? In this paper, the repellent characteristics of current existing superhydrophobic surfaces to hot water (50–80 °C) were investigated. Representative superhydrophobic surfaces from lotus leaves, Teflon, silica–fluoropolymer composites and sol–gel processes were fabricated on silicon wafers and their repellency to hot water was evaluated based on water-contact angle (WCA) measurements and water uptake. These so-called superhydrophobic surfaces, which usually exhibit high repellency to cool water (around 25 °C), however, show remarkably decreased repellency to hot water. This may be attributed to a decrease of surface tension when the temperature of water increases and the lower surface tension of hot water makes it a better “wetting agent” to get into the pores and fissures of rough surfaces rather than bridging them with surface tension. According to the theories of surface wettability, the wetting state of water droplets on superhydrophobic surfaces can be described as Cassie–Baxter and Wenzel states, respectively. Superhydrophobic surfaces lose their high repellency to hot water due to the transition of the wetting state from Cassie–Baxter state to Wenzel state. Surface roughness, which is usually used for improving surface hydrophobicity, however, is found to improve the wettability of a solid surface to hot water. The results in this paper also show that the wettability of a solid surface is governed by both the surface roughness and surface free energy, but the surface energy is more prevailing than the surface roughness in making the surface repel hot liquids. To fabricate hot water repellent fabrics, a nanocomposite of CNTs and Teflon was prepared and applied to commercial fabrics to produce scalding protection clothes. The repellency of the CNTs–Teflon treated fabrics to hot beverages (water, milk, coffee and tea, 50–80 °C) was studied. The spray test shows that although some superhydrophobic surfaces exhibit high repellency to static water they show reduced repellency to dynamic water.

 Please wait while we load your content...
Please wait while we load your content...