Photoinduced reductive cleavage of some chlorobenzylic compounds. New insights from comparison with electrochemically induced reactions
Abstract
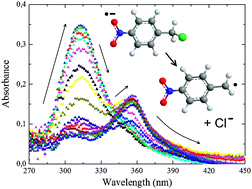
The photoinduced reductive cleavage of the carbon–chlorine bond in some chlorobenzylic nitro- and cyano-substituted compounds has been studied by transient absorption

 Please wait while we load your content...
Please wait while we load your content...