Evaluation of the interchangeability of C–H and C–F groups: insights from crystal packing in a series of isomeric fluorinated benzanilides†‡
Abstract
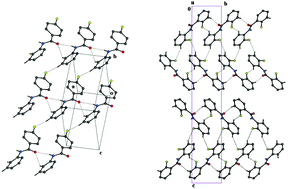
A series of mono- and difluorinated benzanilides have been synthesized and characterized to unravel the importance of interactions involving organic fluorine. The effect of fluorine substitution on the molecular conformation and the solid-state organization of the molecules in the crystalline lattice have been discussed in terms of changes in molecular conformation and the supramolecular aggregation. It is noteworthy that the meta- and para-isomers (mono and difluorinated) are isostructural, with the ortho-isomers possessing minor conformational variations. Furthermore, the molecular conformation as obtained by theoretical DFT calculations is different from the solid-state conformation highlighting the importance of co-operative features brought by N–H⋯O and C–H⋯O hydrogen bonds, along with weak intermolecular interactions involving organic fluorine and aromatic C–H⋯π contacts in the solid state.

 Please wait while we load your content...
Please wait while we load your content...