Recent advances in the semi-pinacol rearrangement of α-hydroxy epoxides and related compounds
Abstract
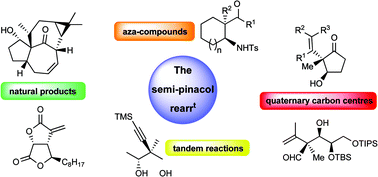
The semi-pinacol rearrangement is fast becoming an extremely reliable reaction in organic synthesis allowing the rapid construction of relatively complex stereodefined products in high yield. Recent advances in asymmetric synthesis have also enabled enantiopure precursors to partake in the rearrangement showing that extremely high levels of stereospecificity are observed. In this critical review recent advances in the semi-pinacol rearrangement over the past 15 years are examined which demonstrate the extremely high utility of this reaction towards the development of structurally diverse organic building blocks (74 references).

 Please wait while we load your content...
Please wait while we load your content...