Air and water stable ionic liquids in physical chemistry
Abstract
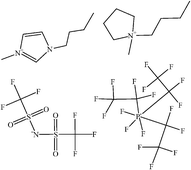
Ionic liquids are defined today as liquids which solely consist of cations and anions and which by definition must have a melting point of 100 °C or below. Originating from electrochemistry in AlCl3 based liquids an enormous progress was made during the recent 10 years to synthesize ionic liquids that can be handled under ambient conditions, and today about 300 ionic liquids are already commercially available. Whereas the main interest is still focussed on organic and technical chemistry, various aspects of physical chemistry in ionic liquids are discussed now in literature. In this review article we give a short overview on physicochemical aspects of ionic liquids, such as physical properties of ionic liquids, nanoparticles, nanotubes, batteries, spectroscopy, thermodynamics and catalysis of/in ionic liquids. The focus is set on air and water stable ionic liquids as they will presumably dominate various fields of chemistry in future.

 Please wait while we load your content...
Please wait while we load your content...