Solid-phase synthesis, characterization and DNA binding properties of the first chloro(polypyridyl)ruthenium conjugated peptide complex
Abstract
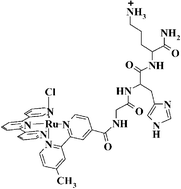
A general method for the synthesis of chloro(polypyridyl)ruthenium conjugated peptide complexes via a solid-phase strategy is described. The method is applied to synthesize two positional isomers of the complex [Ru(terpy)(4-CO2H-4′-Mebpy-Gly-L-His-L-LysCONH2)Cl](PF6). Even though the separation of the isomers was only partially achieved chromatographically, the isomers were unambiguously assigned by NMR spectroscopy. The interactions of the complex [Ru(terpy)(4-CO2H-4′-Mebpy-Gly-L-His-L-LysCONH2)Cl](PF6) with CT-DNA and plasmid DNA, have been studied with various spectroscopic techniques, showing that (i) the complexes coordinatively bind to DNA preferring the bases guanine and cytosine over the bases thymine and adenine after hydrolysis of the coordinated chloride, (ii) electrostatic interactions between the complex cation and the polyanionic DNA chain assist this binding (iii) only in the case of one isomer the peptide does interact further with DNA as evidenced from 31P NMR spectroscopy, (iv) DNA unwinding occurs in all cases with high binding ratio (Ru/base) values (r > 0.3).

 Please wait while we load your content...
Please wait while we load your content...